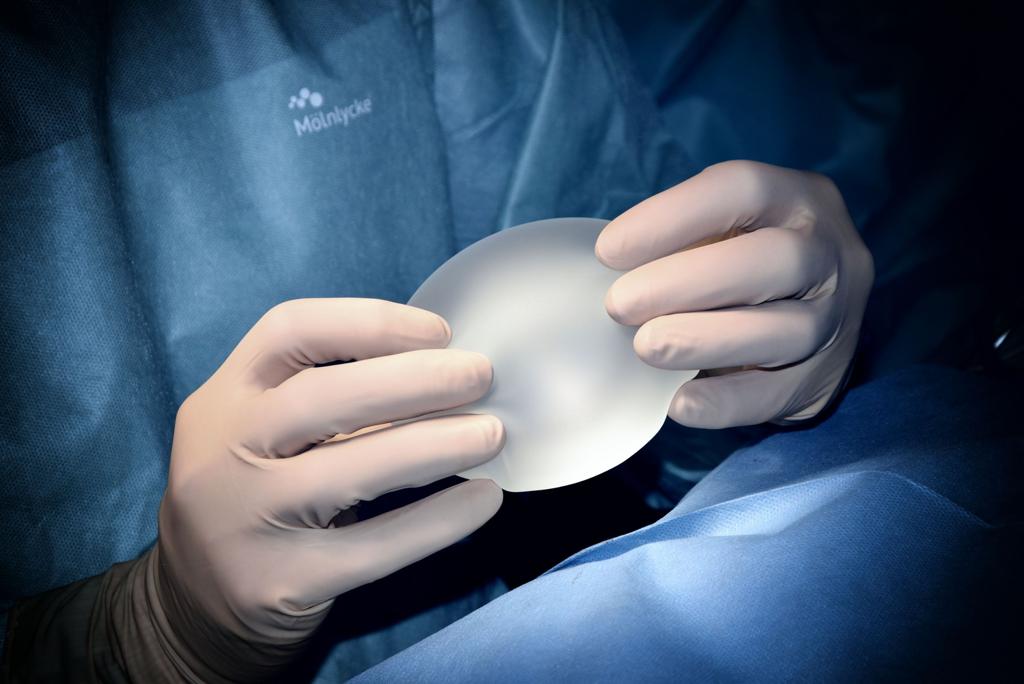
What is BIA-ALCL?
BIA-ALCL is an extremely rare blood cancer (lymphoma) that arises within the capsules of breast implants. As of January 2020, there have been 733 cases reported globally with approximately 78 cases identified in the UK by the end of 2020.
BIA-ALCL is defined, and diagnosed, by the presence of specific molecular markers on the tumour tissue (CD30+) and is differentiated from other forms of ALCL by the absence of anaplastic lymphoma kinase (ALK-). It is thought to arise from a T cell that undergoes malignant change, although the cell-of-origin has not yet been identified.
What causes BIA-ALCL?
The exact causes of BIA-ALCL remain unclear although it is thought to be linked to the surface texturing of breast implants. To date, the majority of cases have been associated with textured implant devices but there is insufficient data to say there is no link between BIA-ALCL and smooth implants. Emerging data suggest that it may be linked to how surface texturing affects your immune response to the implant, but no causative link has yet been identified.
How do I know if I have BIA-ALCL?
BIA-ALCL typically presents 8-10 years after implant insertion and most commonly presents with a collection of fluid around your breast implant (“late seroma”). In the minority of cases, there may also be a lump you can feel. If you are at all concerned about a change in breast shape, volume or new lumps you should consult a specialist for a clinical assessment.
How is BIA-ALCL diagnosed and treated?
BIA-ALCL is diagnosed by analysing the seroma fluid that collects within the capsule. The diagnosis requires the identification of cells that look abnormal and exhibit signature molecular changes (CD30+ALK-) on analysis. The diagnosis can sometimes be challenging, and it is important that histological examination is performed by a pathologist with expertise at diagnosing lymphomas.
BIA-ALCL is very treatable and the survival outcomes after treatment are excellent. In the majority of cases removal of the implant with the implant capsule, en bloc, is sufficient treatment. In cases where there is evidence of spread beyond the capsule, chemotherapy may be required either before, or after, surgery. Your treatment should be undertaken within a specialist centre that has the expertise and experience in dealing with BIA-ALCL cases and has both oncoplastic and haemato-oncology multidisciplinary teams (MDTs). Mr Khan is a member of the multidisciplinary BIA-ALCL team at The Royal Marsden Hospital.
I have textured implants – should I have them removed?
At present, neither the Medicine and Healthcare products Regulatory Agency (MHRA) in the UK nor the Food and Drug Administration (FDA) in the USA have recommended routine removal of textured implant devices and, indeed, the clinical data does not support this policy.
I’m considering having breast implants should I choose smooth rather than textured implants?
The decision making around implant choice, in this regard, can be very challenging and ultimately comes down to a balance of competing risks.
Smooth implants are thought to be less associated with BIA-ALCL, however, there is a higher re-operation rate associated with these devices due to implant migration, rotation and malposition and not all patients may be suitable for a smooth implant. Experience has also shown us that textured devices are easier to control within the breast pocket and are less susceptible to movement. Therefore, the trade-off with choosing a smooth implant to mitigate BIA-ALCL risk is a higher risk of implant movement that may require surgical revision.
It is important to be able to have a transparent discussion about these competing risks in addition to assessing your suitability for a smooth implant when deciding on your implant choice.
For more information on BIA-ALCL or implant options contact Mr Khan’s practice to arrange an appointment.